news
Key indicators of effective dental disinfection
Proper disinfection in dental settings requires monitoring specific parameters that validate system performance. Unlike visual cleanliness assessments, these measurable indicators provide objective data for quality control professionals and safety-conscious consumers.
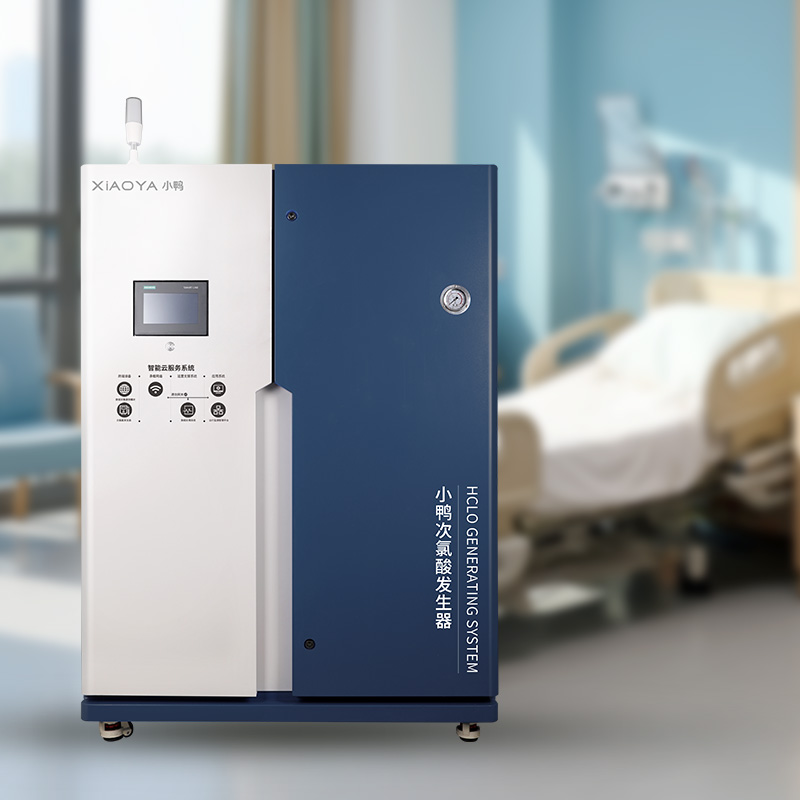
Modern automated systems like the Hypochlorous Acid Generator for Public Health (AQ-P1000) incorporate real-time monitoring of these parameters, eliminating guesswork in disinfection validation. The system's automated alerts notify staff when parameters deviate from optimal ranges.
Microbiological testing protocols
While chemical indicators provide immediate feedback, microbiological testing remains the gold standard for verifying disinfection efficacy. Implement these testing protocols:
- Conduct ATP bioluminescence tests weekly to measure organic residue
- Perform bacterial culture tests monthly for high-risk areas
- Validate sterilization cycles with biological indicators every 6 months
- Document all test results for compliance audits
Common failures in dental disinfection systems
Approximately 30% of dental practices experience disinfection system failures without immediate detection. Understanding these failure modes helps prevent compliance violations and patient safety risks.
Equipment-related issues
Frequent equipment problems include:
- Deteriorating electrodes in hypochlorous acid generators
- Clogged spray nozzles in automated disinfection units
- Sensor drift in monitoring systems
- Inconsistent solution mixing ratios
Automated maintenance alerts and self-diagnostic features in advanced systems can prevent 80% of these common failures through early detection.
Procedural errors
Human factors contribute to 45% of disinfection failures according to recent industry studies. These include:
Automation advantages in disinfection verification
Transitioning from manual to automated disinfection systems reduces human error by 72% while improving documentation accuracy. Modern systems offer three verification layers:
1. Real-time monitoring
Continuous tracking of critical parameters ensures immediate corrective action when deviations occur. Systems should monitor:
- Solution concentration every 15 seconds
- Temperature impacts on efficacy
- Flow rates for proper application
- Cumulative equipment usage hours
2. Automated documentation
Compliance requires maintaining 3-5 years of disinfection records. Automated systems provide:
- Digital logs with timestamps
- Parameter trend analysis
- Alert history documentation
- Exportable reports for audits
3. Predictive maintenance
Advanced systems analyze performance data to predict component failures 2-4 weeks before they impact disinfection efficacy, reducing downtime by 60%.
Frequently asked questions
How often should dental disinfection systems be validated?
Daily operational checks, weekly performance tests, and quarterly comprehensive validations form the industry standard three-tier verification approach. Automated systems may reduce manual testing frequency by 40-60% through continuous monitoring.
What's the most common false positive in disinfection testing?
Residual disinfectants can produce false negative bacterial cultures. Proper neutralization procedures must precede microbiological testing. Automated systems typically include built-in neutralization cycles for accurate testing.
Can automated systems replace all manual disinfection checks?
While automation handles 85-90% of routine monitoring, monthly manual verification remains essential for system calibration and comprehensive audits. The optimal approach combines automated monitoring with periodic human oversight.
Why choose automated disinfection verification?
Modern healthcare facilities require disinfection systems that combine reliability with verifiable performance. Automated solutions offer:
- 24/7 parameter monitoring with 99.7% accuracy
- Reduced labor costs for manual testing
- Compliance-ready documentation systems
- Early warning for equipment maintenance
For facilities considering upgrades, the Hypochlorous Acid Generator for Public Health (AQ-P1000) represents the next generation in automated disinfection verification, combining real-time monitoring with predictive analytics for unparalleled safety assurance.
While automation handles 85-90% of routine monitoring, monthly manual verification remains essential for system calibration and comprehensive audits. The optimal approach combines automated monitoring with periodic human oversight.
Residual disinfectants can produce false negative bacterial cultures. Proper neutralization procedures must precede microbiological testing. Automated systems typically include built-in neutralization cycles for accurate testing.
Daily operational checks, weekly performance tests, and quarterly comprehensive validations form the industry standard three-tier verification approach. Automated systems may reduce manual testing frequency by 40-60% through continuous monitoring.
NEWS
 The most common blind spots in animal husbandry disinfection plans
The most common blind spots in animal husbandry disinfection plans How to evaluate animal husbandry disinfection equipment before purchase
How to evaluate animal husbandry disinfection equipment before purchase How often should animal husbandry disinfection protocols be updated?
How often should animal husbandry disinfection protocols be updated? HClO water treatment for hospitals under stricter hygiene standards
HClO water treatment for hospitals under stricter hygiene standards What causes inconsistent results in HClO hospital water systems?
What causes inconsistent results in HClO hospital water systems?
Leave us a message