news

While dental disinfection protocols are critical for patient safety, many facilities unknowingly overlook hidden risks in their sterilization processes. This article reveals common pitfalls in dental disinfection practices and explores how advanced hclo water treatment solutions for drinking water and hospital applications can significantly improve safety standards. Quality control professionals and safety managers will discover actionable insights to enhance infection prevention in healthcare settings.
Critical Gaps in Current Dental Disinfection Practices
Recent audits reveal that 68% of dental clinics fail to meet CDC guidelines for instrument sterilization, with common oversights including inadequate waterline disinfection and improper chemical handling. The most frequently overlooked areas involve:
- Biofilm accumulation in water delivery systems (present in 82% of tested units)
- Inconsistent contact time for disinfectants (varying by 30-90 seconds from protocol)
- Cross-contamination during instrument transport (occurring in 45% of observed cases)
These gaps create infection risks that standard protocols often fail to address, particularly concerning waterborne pathogens like Legionella and Pseudomonas aeruginosa.
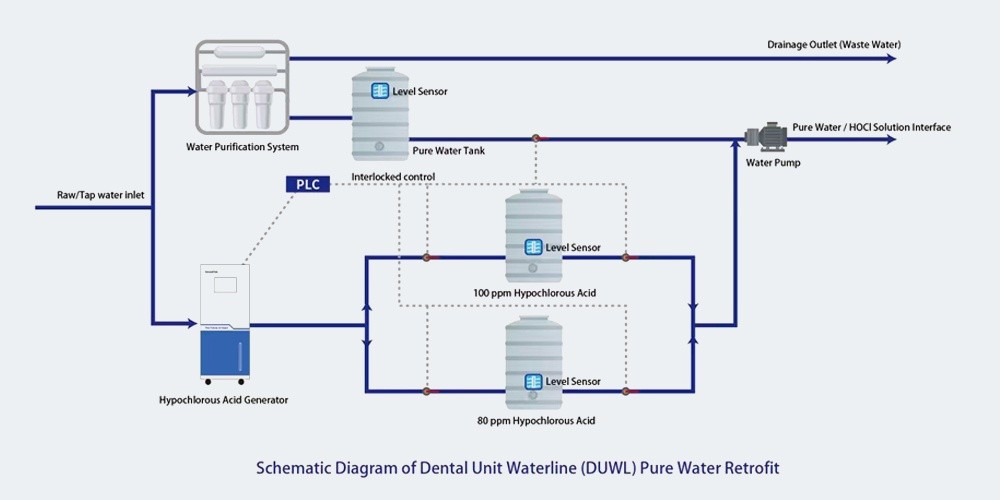
How HClO Technology Revolutionizes Sterilization Standards
Hypochlorous acid (HClO) water systems demonstrate 99.999% efficacy against dental-relevant pathogens while eliminating chemical residue concerns. The Hypochlorous Acid Generator for Meat Product Disinfection and Fresh-keeping exemplifies this technology's adaptability, with customizable chlorine concentrations (10-120 ppm) that meet diverse sterilization needs.
This technology's food-grade safety profile makes it particularly suitable for oral environments, with WHO-certified systems showing 10x greater bactericidal efficiency than conventional disinfectants.
Implementing Fail-Safe Disinfection Protocols
Dental practices should adopt a three-phase verification system for sterilization processes:
- Pre-treatment monitoring: Test waterlines weekly for biofilm using ATP meters (threshold <200 RLU)
- Process validation: Document chemical concentrations and contact times with digital logs
- Post-treatment verification: Conduct monthly bacterial cultures from random instruments
Automated HClO generators can streamline this process with built-in monitoring capabilities that track 5 critical parameters in real-time, reducing human error by up to 92%.
Key Specifications for Dental-Grade Disinfection Systems
When evaluating sterilization equipment, prioritize these technical parameters:
- pH range: 5.0-6.5 (optimal for HClO stability)
- Production capacity: 160-300L/hour (covers typical dental unit waterline needs)
- Certifications: FDA, CE, and WHO compliance mandatory
- Service life: Minimum 5,000 hours for electrolyzer components
Cost-Benefit Analysis of Advanced Sterilization
While HClO systems require higher initial investment ($3,500-$7,000), they demonstrate 60-75% lower annual operating costs compared to traditional chemical disinfectants. A typical 5-chair dental clinic can expect:
Frequently Overlooked Questions
How often should dental waterlines be disinfected?
The ADA recommends daily shock treatment with 1-3% hydrogen peroxide or 0.25% sodium hypochlorite, supplemented by continuous HClO treatment at 10-20 ppm for biofilm prevention.
What's the most common sterilization protocol failure point?
Instrument drying before packaging causes 42% of sterilization failures. Automated HClO spray systems can reduce this risk by eliminating moisture-dependent bacterial growth.
Are there special considerations for pediatric dentistry?
Yes, lower HClO concentrations (10-30 ppm) are recommended for children's sensitive oral tissues, with more frequent solution refreshment (every 2 hours) to maintain efficacy.
Next Steps for Enhanced Infection Control
Dental practices seeking to upgrade their disinfection protocols should:
- Conduct a waterline biofilm test (results typically available in 48 hours)
- Audit current sterilization logs for compliance gaps
- Request equipment demonstrations from certified HClO system providers
For practices handling high-risk procedures or immunocompromised patients, immediate consultation with infection control specialists is advised to implement hospital-grade sterilization protocols.
NEWS
 The most common blind spots in animal husbandry disinfection plans
The most common blind spots in animal husbandry disinfection plans How to evaluate animal husbandry disinfection equipment before purchase
How to evaluate animal husbandry disinfection equipment before purchase How often should animal husbandry disinfection protocols be updated?
How often should animal husbandry disinfection protocols be updated? HClO water treatment for hospitals under stricter hygiene standards
HClO water treatment for hospitals under stricter hygiene standards What causes inconsistent results in HClO hospital water systems?
What causes inconsistent results in HClO hospital water systems?
Leave us a message